MSA: Measurement System Anarchy
Posted: March 26, 2013 Filed under: Creative, Unusual, Amusing, Measurement and Analytics | Tags: Black Belts, kilogram, Lean Six Sigma, measurement, measurement system analysis, MSA, performance improvement, system of measurement, watt balance, weights Leave a commentPerformance improvement professionals and executives like metrics: “what gets measured gets done” they say. But how to properly measure? This is not as easy as it sounds. To understand the issue of measurement at its most fundamental level, look at time, distance and weight.
Time is now defined by readily available clocks that use caesium atoms as their pendulums, while distance is specified in terms of the speed of light in metres per second (the caesium atoms having already provided the value of a second), and these days that speed can be measured with great accuracy using easily purchased equipment. Mass, however, remains stubbornly stuck in Paris, under three concentric glass lids (pictured) strangely reminiscent of cheese covers, which are intended to stop it either absorbing or shedding matter and thus changing in value.
Unfortunately, the cheese covers have not worked. Over the years the standard kilogram has put on weight, or possibly lost it. Nobody quite knows which. But they do know that it is not the same kilo that went under the lids in 1889. Which is wonderful news for writers of newspaper headlines, who have been handed a post-Christmas gift about seasonal weight-gain not being as bad as the bathroom scales might suggest, but is awkward for physicists, who are left unsure what anything in the universe actually does weigh.
The international prototype kilogram (IPK), the technical name for the cylinder in Sèvres, does not in fact keep a constant mass. Over the years pollutants from the air settle on its surface, causing its mass to rise. Attempts to clean it then cause its mass to fall. As a result, what science understands by a kilogram has varied, but has done so in a way that is, by definition, unmeasurable.
The cleaning that attempts to keep the kilogram constant is done by a member of staff at the bureau, using a combination of steam and a chamois-leather cloth soaked in ethanol and ether. But given the sensitivity of modern instruments, which can detect gains or losses of the order of billionths of a kilogram, it must be done properly. Too gentle a rubdown leaves contaminants on the surface. Too vigorous a scrubbing erodes the metal itself.
Worse, the IRK is not the only such cylinder in the world. Dozens of replicas exist in official laboratories in many countries. Each must be cleaned to the same exacting standards if it is to remain a true copy of the French original. In practice, that has often meant shipping the whole lot back to France so that the same person can scrub them all down.
Now, though, Peter Cumpson and Naoko Sano, a pair of researchers at the University of Newcastle, in England, reckon they have come up with a better way. In a paper just published in Metrologia, they describe the results of a method that combines traditional washing with a technique adapted from the chip-manufacturing business, which employs a combination of ultraviolet light and the ozone that such light synthesises from the air to strip pollutants away.
The idea of using ultraviolet light was first proposed back in the 1990s, says Dr Cumpson, but has yet to be adopted (“metrologists”, he notes, “are—rightly—rather conservative people”). This latest paper, which reports the results of using the method on strips of platinum-iridium alloy (the IPK and its replicas being far too precious for experimentation), shows that the method does a good job of removing the carbon-based gunk that settles onto the weights from the air. Importantly, says Dr Cumpson, unlike a manual rubdown, his procedure can easily be standardised, and the kit necessary to perform it is cheap and widely available, allowing laboratories around the world to keep their own kilogram replicas clean.
It is not, however, perfect. Besides atmospheric muck, researchers reckon that other things are altering the masses of the world’s kilograms. One significant effect, says Dr Cumpson, is contamination with mercury. The likely source is not atmospheric pollutants, but lab accidents. “Say you drop a thermometer and it shatters,” he says. “A couple of blobs of mercury fall through the floorboards and evaporate slowly over the years. Some of that mercury is then deposited [on the kilograms]”. Unlike the atmospheric junk, which simply encrusts the surface, mercury seeps into microscopic flaws in the cylinders’ surfaces created by the polishing process. “Once it gets in, it’s in there for good. You can’t remove it,” says Dr Cumpson.
Science would thus love to be free of this awkward lump of metal, but attempts to define mass objectively—with reference to, say, the mass of a proton—have always foundered on the question: “So how do you measure that?” For all the fancy equipment that scientists now have for monitoring the behaviour of caesium atoms and the value of the speed of light, no one has come up with a more accurate way of measuring mass than taking the Parisian ingot out of its sarcophagus from time to time, and putting it on a set of scales.
Some physicists do believe they have an answer. It involves Planck’s constant (the fundamental principle of quantum physics), the speed of light and an apparatus called a watt balance. If a meeting next year approves this idea, the Parisian mass will at last become redundant. But the watt-balance approach itself has critics, for such balances are costly.
According to the Bureau International des Poids et Mesures (http://www.bipm.org/en/scientific/elec/watt_balance/) “when the international prototype of the kilogram (IPK) was manufactured, another six official copies with exactly the same characteristics were also produced. The mass of each official copy has been compared to the mass of the IPK on three separate occasions since 1889 (see the graph below). The average mass of the official copies seems to increase over time with respect to the mass of the IPK, with a drift of approximately 50 µg in 100 years.”
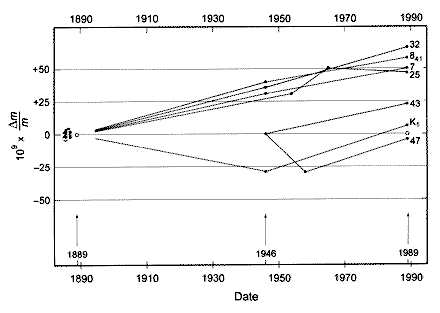
Relative mass differences between each of the six official copies and the IPK. These mass comparisons have taken place on three occasions since 1889. The mass of the IPK defines the zero value of the vertical axis of this graph.


